Carcinoembryonic antigen, monoclonal, by IHC-12376 - Technical only, 12379 - Technical & interpretation
Test info
Carcinoembryonic antigen, monoclonal, by IHC
12376 - Technical only, 12379 - Technical & interpretation
LAB12376
LAB12379
LAB12379
CEA, monoclonal
- All IHC stains will include a positive control tissue
- 95% of adenocarcinomas of colon, lung, breast, pancreas, biliary tract are CEA positive
- Renal cell, prostatic, hepatocellular, adrenocortical, and thyroid carcinomas as well as mesotheliomas are usually negative for CEA
NOTE: When CEA is ordered, the monoclonal CEA will be automatically performed unless the polyclonal antibody is specifically requested. Polyclonal CEA also reacts with NCA (nonspecific cross reacting antigens) and BGP (biliary glycoproteins). It is the unique CEA/BGP expression that is useful in identifying hepatocellular carcinomas (see section on immunohistochemistry panels). Thus, if the hepatocellular carcinoma panel is requested, then both monoclonal and polyclonal CEA will be automatically performed.
Specimen
Tissue
Prepare a formalin-fixed, paraffin-embedded (FFPE) tissue block
Formalin-fixed, paraffin embedded (FFPE) tissue block
FFPE tissue section mounted on a charged, unstained slide
Ambient (preferred)
- Unlabeled/mislabeled block
- Insufficient tissue
- Slides broken beyond repair
Performance
AHL - Immunohistochemistry
Mo - Fr
1 - 2 days
Immunohistochemical staining and microscopic examination
Clinical and Interpretive info
If requested, an interpretive report will be provided
Specifications
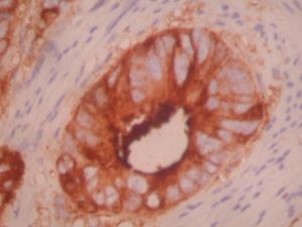
- CEA is a family of HMW glycoproteins
- Expression of CEA by tumor is often proportional to degree of differentiation
- CEA also labels PMNs
Staining pattern
- Cytoplasmic and apical based staining
Billing
88342 - 1st stain
88341 - each additional stain
88341 - each additional stain
Tracking
06/19/2017
10/17/2018
01/19/2026