Immunohistochemistry
Director: Lori Ryan, MD
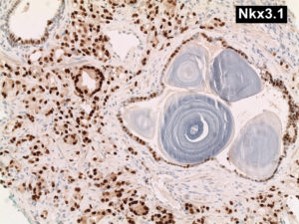
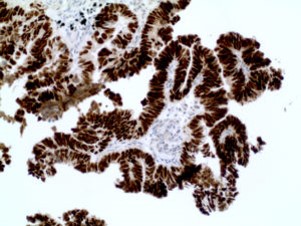
NKx3.1 Cytokeratin 18
The Allina Health Immunohistochemistry (IHC) Laboratory provides immunohistochemical and in situ hybridization (ISH) staining on slide based tissue specimens. It serves as a reference laboratory for clinical outreach work and as a resource for clinical research projects.
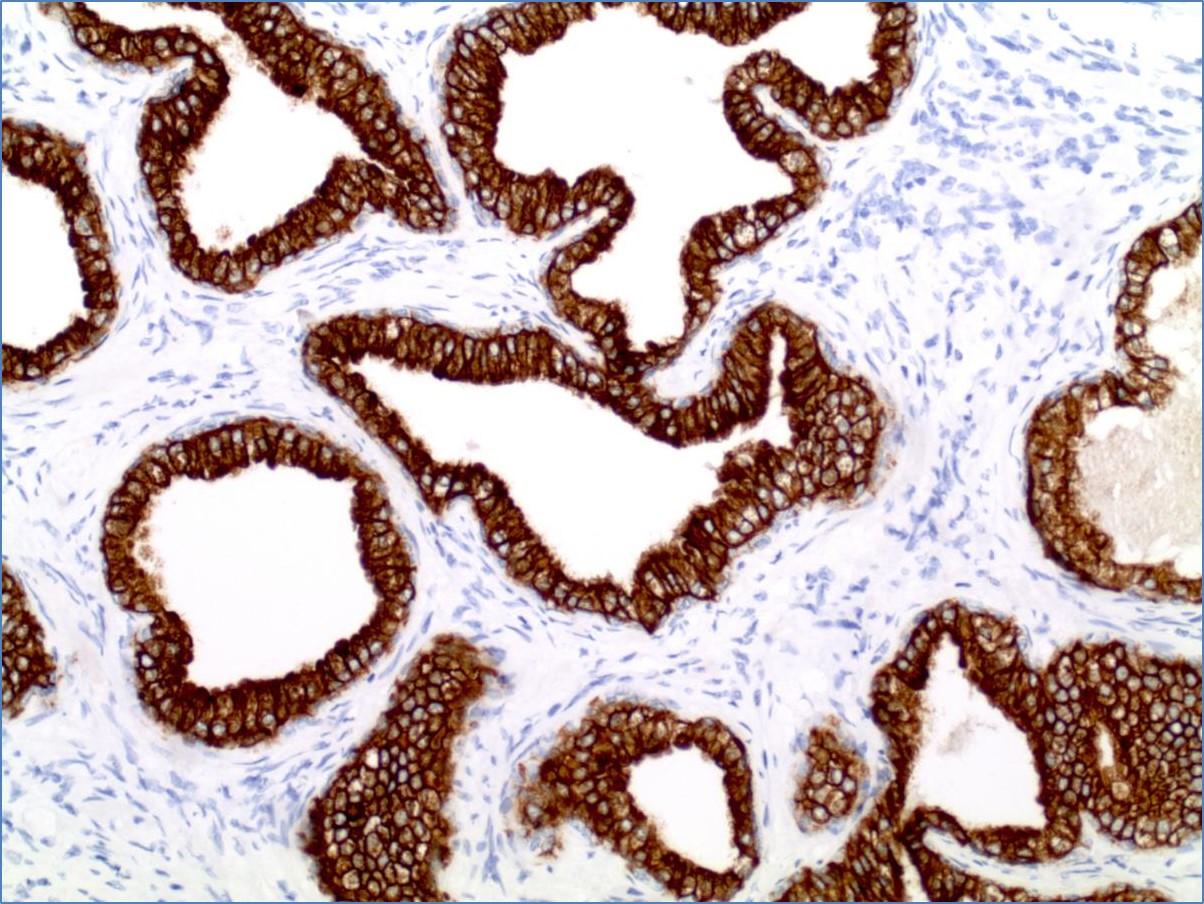
Immunohistochemical methods are useful in determining the cell lineage of tumors, to compliment the histologic assessment in reaching a clinical diagnosis. This information may provide prognostic information regarding malignant tumors by analyzing receptor status, protein production, and proliferation potential of a particular tumor. ISH is a highly sensitive analysis that evaluates for RNA and DNA transcripts within cells. This technique can determine the presence of light chain immunoglobulins, which are extremely important in the evaluation of benign and malignant plasmacytic and lymphoid proliferations. ISH testing can also be used to identify certain viral infections that may have clinical implications, significant for patient prognosis. These studies can be performed on paraffin-embedded tissue or cell blocks.
The Allina Health IHC laboratory staff is highly trained and performs complex testing utilizing state of the art equipment and reagents. All testing is automated using either the Leica Bond or the Ventana Ultra instrument platforms. Stringent quality control measures are utilized throughout the testing processes. All antibody clone evaluations, procedures, titers and digestions have been carefully optimized under the direction of pathologists with subspecialty training.
IHC evaluation can be requested with professional interpretation by a Hospital Pathology Associates (HPA) pathologist. Additionally, most IHC stains are also available as “technical only” orders, allowing the originating pathologist to interpret and bill for the IHC interpretation. If a paraffin block is provided by the requesting laboratory, all IHC stains will include a positive control tissue with each case, frequently on the corresponding patient’s slide(s).
Routine clinical testing is performed on a daily basis (Mo - Fr), and slides (and results) are typically available within 24 hours.
A listing of currently provided testing, with links to complete information in our test catalog, can be found here.
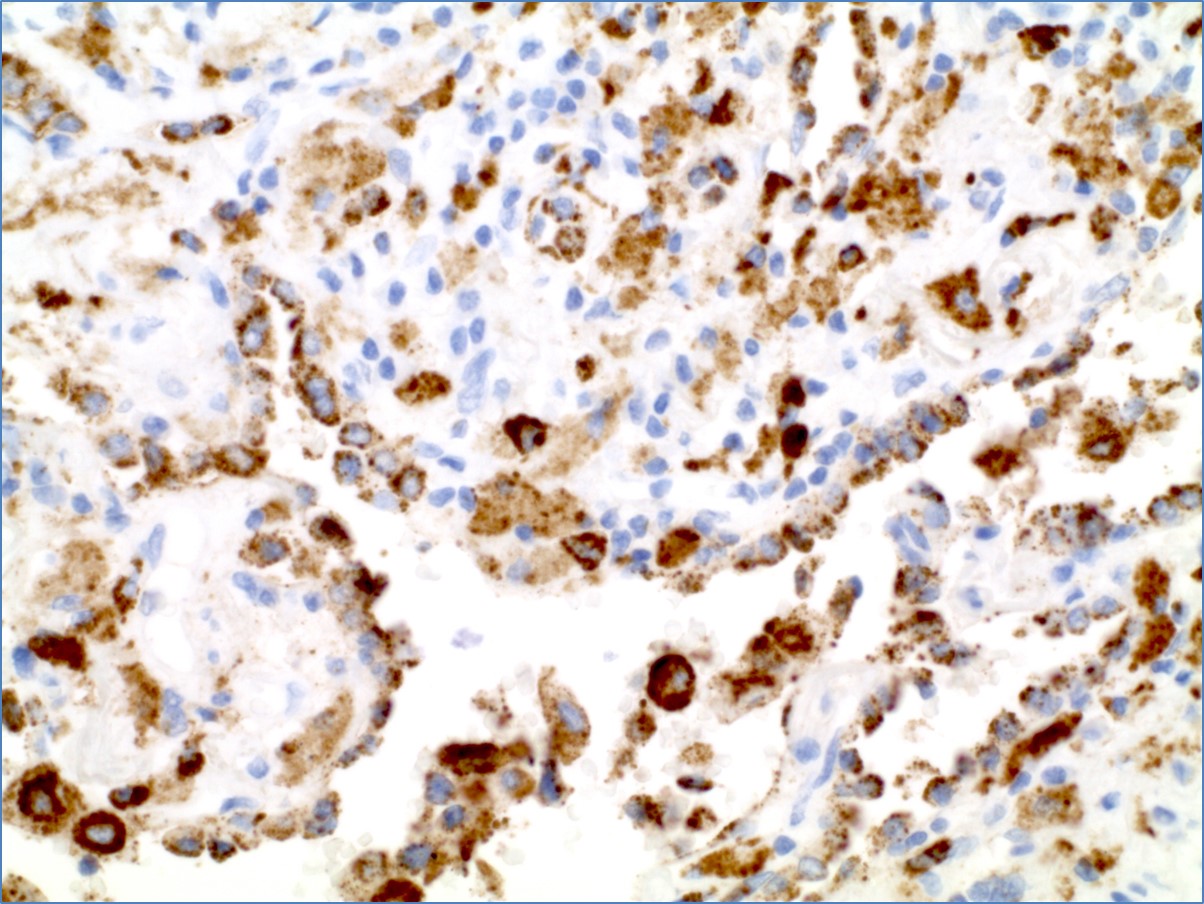
Napsin A PAX8