Cytomegalovirus (CMV) inSIGHT™ T Cell Immunity testing
Cytomegalovirus (CMV) inSIGHT™ T Cell Immunity testing-994
Due to sample stability restrictions, Pathologist review is required before sample is drawn.
The CMV inSIGHT™ T Cell Immunity Panel measures the strength of T cell responses to Cytomegalovirus (CMV) specific antigens. It evaluates and reports the activity of CD4 and CD8 T cell responses independently. Effective T cell immunity against CMV is a factor in controlling CMV viral latency. CMV can affect patients with weakened immune systems and is a common risk factor in patients following solid organ or hematopoietic stem cell transplant.
3.0 mL (pediatric)
- Collect Monday through Friday only
- Immediately following collection, mix sample thoroughly by gently inverting 8 - 10 times, to prevent clotting
- Tube must be at least ¾ full to maintain proper ratio of blood to anticoagulant
- Do not spin
- Submit original tube
- Send ambient overnight same day
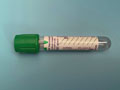
Dk green Sodium heparin (Na hep), no gel
Ambient (strict) - 32 hours
Refrigerated - NO
Frozen - NO
Specimen must be transported to the Allina Health Central Laboratory, in a STAT bag, on the day of collection. Specimen must arrive at AHL by 1:00 pm to facilitate shipping to the performing laboratory.
- Whole blood received after stability (32 hours after collection)
- Whole blood received cold or frozen
- Tubes received less than 3/4 full
- Specimens received in lithium heparin, ACD or EDTA anticoagulants.
Flow Cytometry
See report