Middle East Respiratory Syndrome Coronavirus (MERS-CoV)
Middle East Respiratory Syndrome Coronavirus (MERS-CoV)-994
For patients with clinical suspicion of MERS-CoV, please call MDH at 651-201-5414, or toll-free at 1-877-676-5414, to obtain testing approval and consultation on specimen collection.
The CDC recommends the collection of specimens from lower respiratory tract, upper respiratory tract and blood. At a minimum, MDH recommends the collection of a repiratory specimen (upper or lower) and a blood specimen.
NP/OP swab collection
NP/OP specimens need to be combined, placing both swabs in the same VTM vial.
- Use only synthetic fiber swabs with plastic shafts.
- Do not use calcium alginate swabs or wooden shafted swabs.
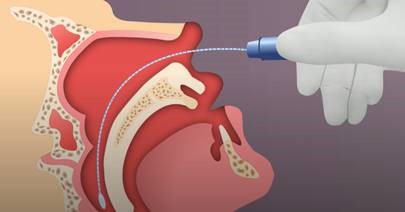
Nasopharyngeal (NP) swab collection:
- Tip the patient’s head back.
- Gently insert the NP swab into the nostril parallel to the palate (not upwards) until resistance is encountered or the distance is equivalent to that from the ear to the nostril of the patient, indicating contact with the nasopharynx.
- If any resistance is met in the passageways, do not force the swab; back off and try reinserting it at a different angle, closer to the floor of the nasal canal, or try the other nostril.
- Gently rub and roll the swab for 10-15 seconds while the swab is in contact with the nasopharyngeal wall.
- The CDC recommends leaving the swab in place for several seconds to absorb secretions.
- Slowly remove the swab and place in the transport medium.
- Break the swab shaft so that it fits into the medium container and recap tightly.
- Label the specimen appropriately. Document the source “NP” on the label.
New England Journal of Medicine video on NP swab collection: Nasopharyngeal Swab Collection Video
Gold serum separator (SST) tube:
- Allow sample to clot for a minimum of 30 minutes
- Spin within two (2) hours of sample collection
Serum:
Gold SST
Upper respiratory swab:
NP/OP swab sumbitted in Viral Transport Media (M4 or M5)
Sputum
Brocheoalveolar lavage
Bronchial wash
Tracheal aspirate
Pleural fluid
Upper respiratory specimen:
Nasopharyngeal and oralpharyngeal swab (NP/OP swab)
Lower respiratory specimen:
Sterile container
Upper respiratory specimen:
Viral Transport Media (M4 or M5)
Refrigerated (preferred) - 72 hours
Frozen (OK)